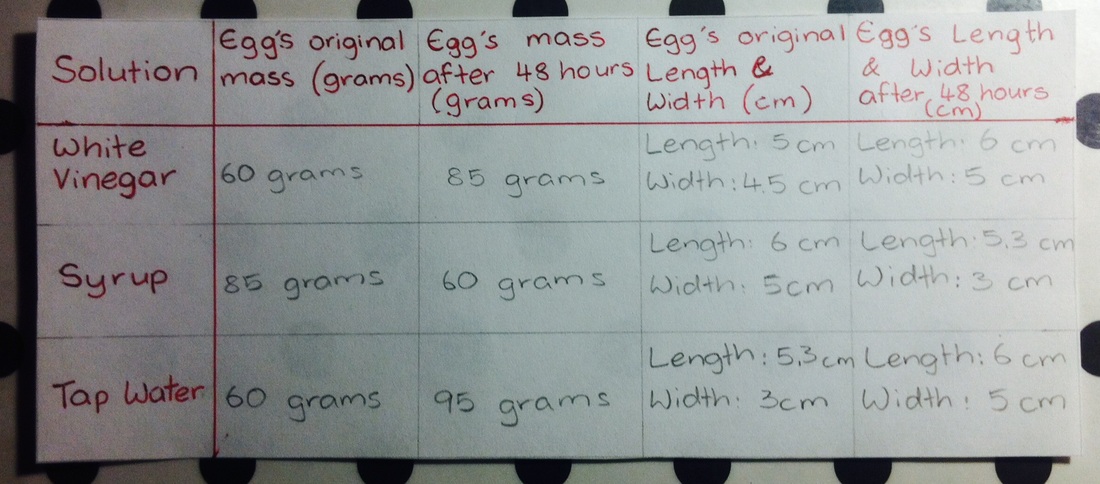
Eggsperiment Results Table
The result is that the egg increased in size and mass when placed in white vinegar solution and tap water solution. But decreased in size and mass when placed in syrup.
Eggsperiment Discussion - All related images are shown on the 'Home' page
Experiment 1 - The Eggsperiment with Vinegar solution
The first experiment conducted was the egg placed in vinegar solution which allowed the egg to become soft and bouncy like jelly. Vinegar is a weak acid which is 5% acetic acid in water (meaning vinegar is largely just water). The egg has a shell that is made up of calcium carbonate and has a lot of proteins and fats dissolved in it, resulting to vinegar to have a lower solute concentration. When vinegar combines together with the egg's shell, the calcium carbonate is eaten away or break down by the vinegar, which then it produces carbon dioxide. The egg then reveals a semi-permeable membrane where it allows small molecules like water to enter it. While this action is taken place, more water is entering the egg instead of leaving it. This action is call osmosis which is the movement of water through a semipermeable membrane from a weak solution to a strong solution.
Experiment 2 - The Eggsperiment with Syrup solution
The second experiment conducted was the egg placed in syrup solution which allowed the egg to become deflated. Osmosis takes action once again in this experiment like the first one in order to shrink the egg. Syrup is a sugary substance that has a solute concentration much greater than the egg.
Experiment 3 - The Eggsperiment with Tap Water solution
The third experiment conducted was the egg placed in tap water solution which allowed the egg to became inflated again. Osmosis takes place in this experiment again too and the egg increases in size and weight more than the first and second experiments. Tap water is a weaker solution than the other solutions used in the first and second experiment so it was able to pass through the eggs semi-permeable membrane more easily.
The first experiment conducted was the egg placed in vinegar solution which allowed the egg to become soft and bouncy like jelly. Vinegar is a weak acid which is 5% acetic acid in water (meaning vinegar is largely just water). The egg has a shell that is made up of calcium carbonate and has a lot of proteins and fats dissolved in it, resulting to vinegar to have a lower solute concentration. When vinegar combines together with the egg's shell, the calcium carbonate is eaten away or break down by the vinegar, which then it produces carbon dioxide. The egg then reveals a semi-permeable membrane where it allows small molecules like water to enter it. While this action is taken place, more water is entering the egg instead of leaving it. This action is call osmosis which is the movement of water through a semipermeable membrane from a weak solution to a strong solution.
Experiment 2 - The Eggsperiment with Syrup solution
The second experiment conducted was the egg placed in syrup solution which allowed the egg to become deflated. Osmosis takes action once again in this experiment like the first one in order to shrink the egg. Syrup is a sugary substance that has a solute concentration much greater than the egg.
Experiment 3 - The Eggsperiment with Tap Water solution
The third experiment conducted was the egg placed in tap water solution which allowed the egg to became inflated again. Osmosis takes place in this experiment again too and the egg increases in size and weight more than the first and second experiments. Tap water is a weaker solution than the other solutions used in the first and second experiment so it was able to pass through the eggs semi-permeable membrane more easily.
Eggsperiment Conclusion
In conclusion, the aim 'to determine why the egg changes in size and weight when placed in three different types of liquid solutions' has been solve. The hypothesis which was 'The naked egg when placed in tap water solution will go back to it's original size and weight at the start of the first experiment. But change in weight and size when placed in syrup solution and vinegar solution.' was nearly exact. Sadly the hypothesis was wrong in saying that the egg will go back to it's original size from the start of the first experiment.